THIS WEBSITE USES COOKIES
We use cookies to personalise content, to provide social media features, and to analyse our traffic. By choosing 'allow all cookies', you consent to our cookies.
To find out more, read our privacy policy and cookie policy.
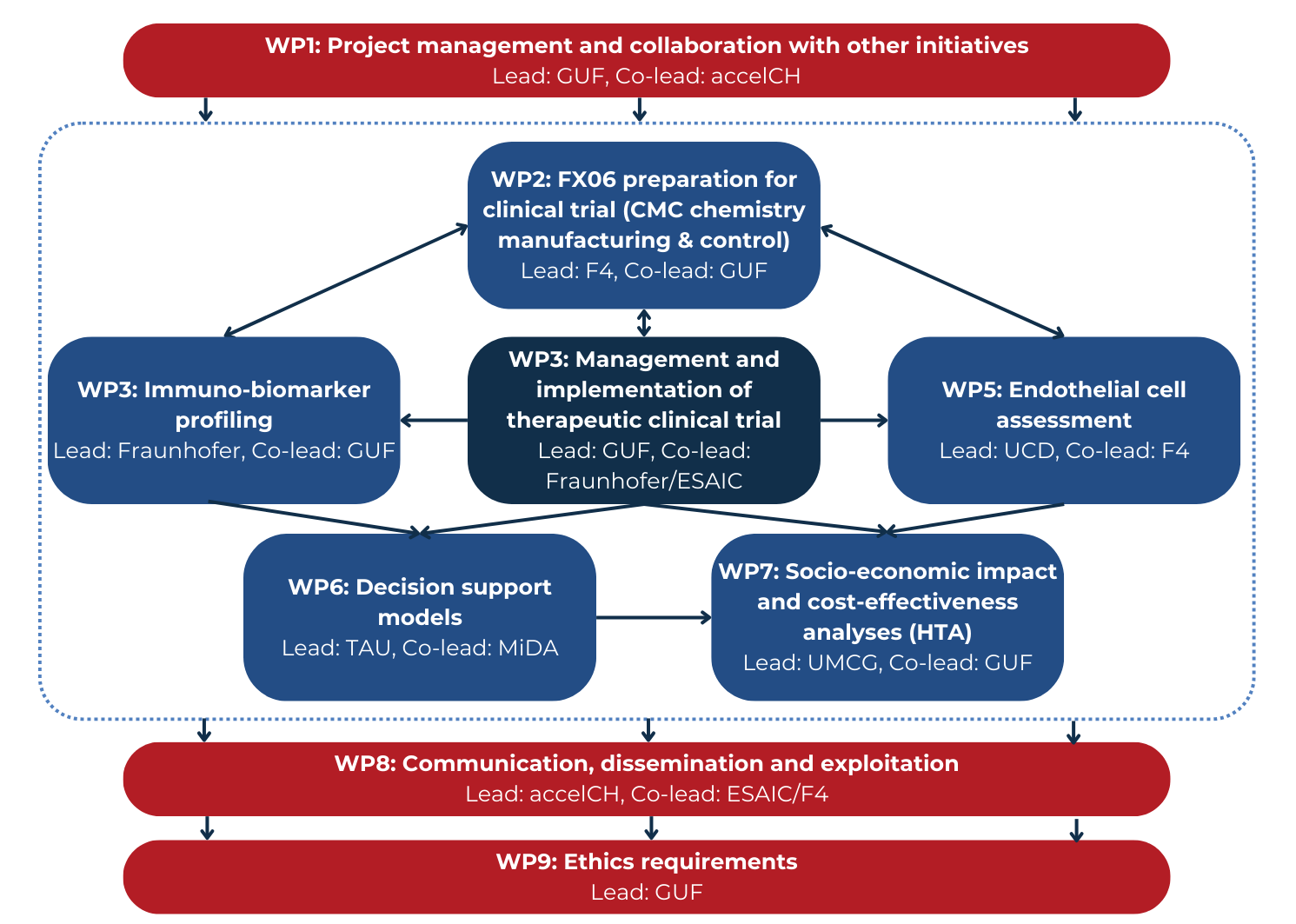
WP1 will be coordinated and managed by GUF and supported by executive committees and board members. WP1 will ensure that COVend’s objectives are achieved and the collaboration with other initiatives are added value to the project.
WP2 and WP3 focus on the preparation and the testing of the promising therapeutic candidate FX06.
WP4, WP5 and WP6 are complementary in that they develop tools (biomarker profile incl. endothelial cell behaviour and AI-based predictive models for decision support) to prove and enhance the benefits of FX06 and thereby offer an individualised therapy.
WP7 will develop the early HTA model to assess the cost effectiveness and the social and economic implications of the new therapy.
WP8 will ensure proper dissemination of project results to expert and non-expert audiences (i.e., researchers, healthcare funders, patients, patient advocacy groups and policy makers).
Lead: GUF
The main objectives of WP1 are to ensure an efficient and effective project management. It will:
Lead: F4
The main goal of WP2 is to set up and perform all prerequisites necessary for the conduction of a large scale ph IIb/III clinical study in moderate COVID-19 patients in several European countries. More specifically, we will:
Lead: GUF/Fraunhofer
The main goal of WP3 is to conduct the multicentric clinical study in several European countries to provide data regarding safety and efficacy of FX06 in the treatment of patients with mild/moderate ARDS related to COVID-19 or other aetiologies. More specifically, we will:
Lead: Fraunhofer/GUF
Applying multiomic technologies, we will provide information about plasma protein levels requested as secondary endpoints in WP3. Further, we aim to identify and follow up on major biomarkers in the blood of patients treated with FX06 versus placebo over time (longitudinal). Newly identified molecules involved in FX06 activity will be validated in WP5. The omics-derived data will be analysed in WP6 by machine learning and AI and integrated into modelling approaches. More specifically, we will:
Lead: UCD
There is accumulating evidence that SARS-CoV-2 does not only infect pulmonary epithelial, but also endothelial cells (ECs). The endothelium is also involved in thrombotic events and acute pulmonary embolism associated with COVID-19. Endothelial damage is key in facilitating fatal COVID-19 and protecting the vasculature may be critical for tackling the acute respiratory infection, organ failures (heart, liver, kidney) and thromboembolism, which are the most severe complications of COVID-19. FX06 does not belong to any known drug class and targets the host response via the endothelium, which is affected directly and indirectly by the virus. FX06 may be an innovative promising therapy to improve endothelial dysfunction, capillary leak, arterial oxygenation, and lung function and, thus possibly able to improve disease severity and prevent progression. In the restructured clinical study, we will investigate the molecular mechanism of FX06. Specifically, we aim:
Lead: TAU
The main goal of WP6 is enable and carry out data analytics and modelling towards personalised decision support for FX06 in treatment of mild to moderate ARDS related to COVID-19 or other aetiologies. WP6 interacts closely with WP3 and WP4, using their collected data to generate new information to create multi-variate / multi-modal profiles of patients, study possible side effects and patient state dynamics over time. WP6 will deliver knowledge, and implement this in algorithms, that serve as basis for decision support for personalised FX06-based intervention planning. Furthermore, the results of WP6 will feed into the activities of WP7, allowing to access the (cost-effectiveness) of delivered decision support approaches. Specifically, we will:
The main goal of WP7 is to evaluate FX06 from a health economic perspective and identify relevant policies and barriers affecting feasibility of implementation. Given the clinical benefit that will result from the clinical study, can FX06 be considered good value for money within a hospital, within the healthcare system, and for society as a whole? Cost-effectiveness will be determined relative to relevant comparators in close collaboration with WP2 and EMA. More specifically, we will:
Lead: accelCH
The main goal of WP8 is to raise awareness about COVend and to achieve a wide outreach of the project results. We will:
Lead: GUF
The main goal of WP9 is to to ensure compliance with the ‘ethics requirements’ set out in this work package

Funded by the European Union. Views and opinions expressed are however those of the author(s) only and do not necessarily reflect those of the European Union or the European Health and Digital Executive Agency (HADEA). Neither the European Union nor the granting authority can be held responsible for them.
FOR INQUIRIES, PLEASE SEND US AN EMAIL AT [email protected]
© accelopment Schweiz AG · [email protected] · ISO 9001 certified · Read our Privacy Policy